 
Similar SLLS glass formulations have recently been demonstrated at the Oak Ridge Reservation (ORR) in a full scale melter with mixed (radioactive and hazardous) wastes.The low melting temperatures achieved with the SLLS glass minimize volatilization of hazardous species such as arsenic, lead, and selenium during vitrification. The SRTC soda-lithia-lime- silica (SLLS) glass melted at 1050 degrees C. Phase separation is known to compromise waste glass stability. 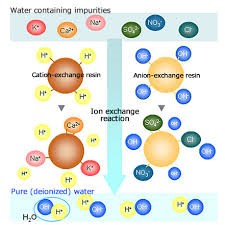
Lime, MgO, BaO and PbO are all constituents of the FEMP wastes and thus subject to phase separation when vitrified in borosilicate glass. 3.2).C is a countercation (for example, Na +, H +), which is mobile and able to be exchanged with other cations found in water. In the case of cation-exchange membranes, permanently fixed ions carry negative charge (Fig. Resins are usually made from the polystyrene polymer backbone and differ. The results of this study provided useful information on the utilization of ion exchange resins as release modifiers in matrix systems.= ), where (MO = CaO, MgO, BaO, and PbO). Cation-exchange resins having formula of R C, where R is a polymer resin matrix with a rigidly fixed ion on its structure. Ion exchange resins consists of two main types, i.e., cation exchange resins, that exchange positively charged ions, such as sodium, for calcium, and anion exchange resins, that exchange negatively charged ions, such as chloride, for arsenic.  
The bulk density as installed in a column includes a normal 35-40. EC-based matrices containing either resin displayed a propensity for disintegration caused by swelling and wicking (water adsorption) actions by the resin. In the water- swollen state, ion exchange resins typically show a specific gravity of 1.1-1.5. Heterogeneous ion-exchange membranes with useful properties contain 6468 wt. This approach is more effective towards eliminating emerging short-chain PFAS which are not removed by carbon-based adsorption processes. Due to the spherical symmetry of granular ion-exchange resins and due to the fact that they are not physically constrained in use, there is generally no functionally important physical damage to the resins from drying and rewetting or from changes of. How ion exchange resins work The resins are prepared as spherical beads 0.5 to 1. Adsorption and ion exchange are considered together because they share many common features, including design concepts and, in some cases, operating cycles, although ion exchange operating cycles are somewhat more complex. The ion exchange (IX) process for PFAS removal is an efficient technology for the remediation of PFAS-laden surface, ground and effluent wastewaters. Water - Structure and Physical Properties. However, Am64 could retard drug release under simulated gastrointestinal conditions. These two kinds of resin are still the most commonly used resins today. Discuss briefly demineralization and deionization of water by ion exchange resins. Such materials are heterogeneous in the sense that their physical properties vary. Dow88 strongly dissociated and lowered the drug release to a greater extent than Am64, which was weakly dissociated. At the same time, the study revealed a preponderance of ion-exchange resins. In deionized water, both resins lowered drug release from HPMC-based matrices by virtue of the gelation property of matrix former and the drug exchange property of embedded resin, in other words in situ resinate formation. In contrast, the addition of Am64 increased matrix hardness and maintained the original thickness, diameter, and friability. The addition of Dow88 to the matrices decreased matrix hardness and increased thickness, diameter, and friability. The matrices were prepared by direct compression of Methocel K4M (HPMC) or Ethocel 7FP (EC) polymeric matrix formers and contained diphenhydramine hydrochloride as a model drug. This study focused on investigating and comparing between the effect of the strongly cationic exchange resin, Dowex 88 (Dow88), and the weakly cationic exchange resin, Amberlite IRP64 (Am64), on the physical properties of matrices and their drug release profiles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
